The centromere/kinetochore plays a fundamental role in accurate chromosome segregation during mitosis and meiosis in eukaryotes (Figure). Its functions include sister chromatid adhesion and separation, microtubule attachment, chromosome movement, and mitotic checkpoint control. Although chromosome segregation errors cause genetic diseases including some cancers, the mechanism by which kinetochores interact with microtubules of the spindle apparatus during cell division is not fully understood.
To understand kinetochore assembly and function in higher vertebrate cells, we are taking multidisciplinary approaches including molecular genetics, cell biology, biochemistry, structural biology, and mouse genetics.
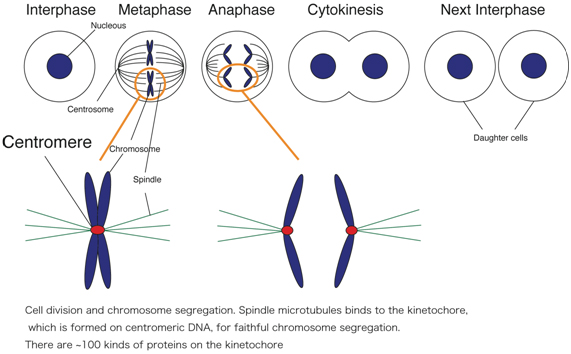
We have been identifying and characterizing kinetochore proteins. We also characterized centromeric DNA. The long-term goal in our laboratory is to understand how kinetochores are formed and how kinetochores function during mitosis. Our research will contribute to a better understanding of human health because chromosome abnormalities, such as aneuploidy and translocations, are tightly associated with tumor development and birth defects.
Currently, we are studying on
- Centromere specification
- Structure and function of kinetochore complexes
- Chromosome segregation during embryogenesis in mice
1. Centromere specification
Centromere region is not specified by specific-sequence information, because centromere DNA is not conserved. Recent idea is that centromere is specified by a sequence-independent epigenetic mechanism. We characterized centromeric DNA in chicken and demonstrated that centromeric DNA sequence from each chromosome contains specific sequence and there is no common sequence, supporting the idea that centromere is specified by an epigenetic mechanism. In addition, we established an experimental system by which we conditionally remove a centromere from the chromosome Z using genetic engineering. Using the experimental system combined with another chromosome-engineering method such as LacO-LacI system, we try to understand how centromere is specified and formed after removal of the endogenous centromere.
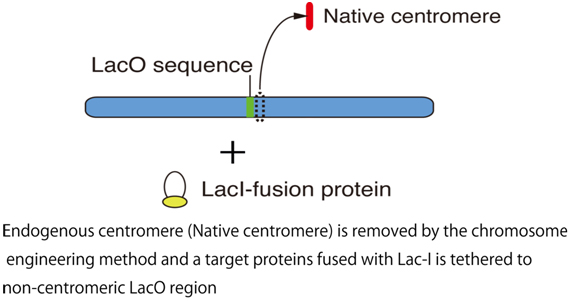
As shown in the Figure, we tether a target protein into the non-centromeric LacO region after removal of endogenous centromere and seek for target proteins, which are responsible for new kinetochore formation.
We also try various experiments to create artificial kinetochore, efficiently. It is necessary to use cell culture, molecular biology, microscopic observation, and genome analysis including ChIP-seq for achievement of this project.
2. Structure and function of kinetochore complexes
The kinetochore is a large multi-protein complex, which consists of ~100 kinds of protein. As the kinetochore is formed on centromeric DNA and binds to microtubules, there are DNA binding proteins and microtubule binding proteins in kinetochore proteins. Although many kinetochore components had not been identified prior to 2000, most of kinetochore proteins have been identified by various proteomic approaches in 2000’. We have identified many proteins, which constitutively localize to kinetochore throughout the cell-cycle. These proteins are called as “CCAN” (Constitutive Centromere Associated Network). Now, there are 16 CCAN proteins. We are studying on structure and function of CCAN proteins.
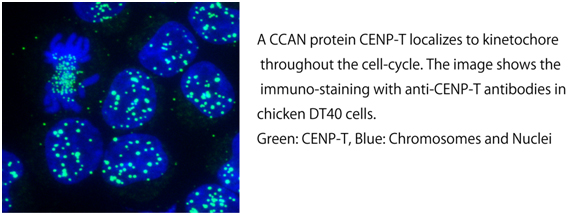
We demonstrated that CCAN is divided into several sub-complexes. We characterize function of each sub-complex in vivo and try to reconstitute each sub-complex in vitro. If we can reconstitute all sub-complexes, we would reconstitute whole CCAN complex. Ultimately, we would create functional kinetochore complex by in vitro reconstitution.
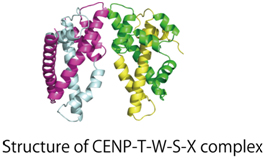
We conduct X-ray crystal structure analysis on the reconstituted sub-complexes. We reported the structure of the CENP-T-W-S-X complex.
It is necessary to take various approaches including cell culture, molecular biology, cell biology, structural biology, and biochemistry for achievement of this project.
3. Chromosome segregation during embryogenesis in mice
We usually use cultured-cells to study kinetochore function in higher vertebrate cells. As chromosome segregation must occur when cells are divided, it is suitable to use cultured-cells for understanding of basic mechanism of kinetochore structure and function. However, times for completion of chromosome segregation are different in various cell types. It is well known that rapid chromosome segregation occurs in cells during differentiation. Therefore, we are interesting about specific mechanisms of chromosome segregation in each cell-type. To perform this project, we are using mice. We recently found that kinetochore composition of cells before differentiation is different from that of cells after differentiation.
We are also making knockout mice of some kinetochore proteins to understand carcinogenesis due to defects of kinetochore function.
It is necessary to use techniques of creation of knockout mice and cytology for this project.












