Organismal Biosystems Laboratories
Laboratory for Embryogenesis
 Prof. SASAKI Hiroshi
Prof. SASAKI Hiroshi
Keywords:
Intercellular communications, Embryogenesis, Mouse, Cell competition, Hippo signaling
Understanding the intercellular communications that support correct development
Our body is made up of numerous cells. Correctness is one of the important characteristics of embryonic development. To achieve such correctness during development, it is required that cells communicate with their neighbors and coordinate their behavior. To understand the mechanisms of such intercellular communications and their roles in embryogenesis, we are focusing on the Hippo signaling pathway, which is involved in intercellular communications through cell-to-cell adhesion, and the cell competition, an intercellular communication by which cells recognize the conditions of their neighbors and eliminate the cells with relatively lower fitness.
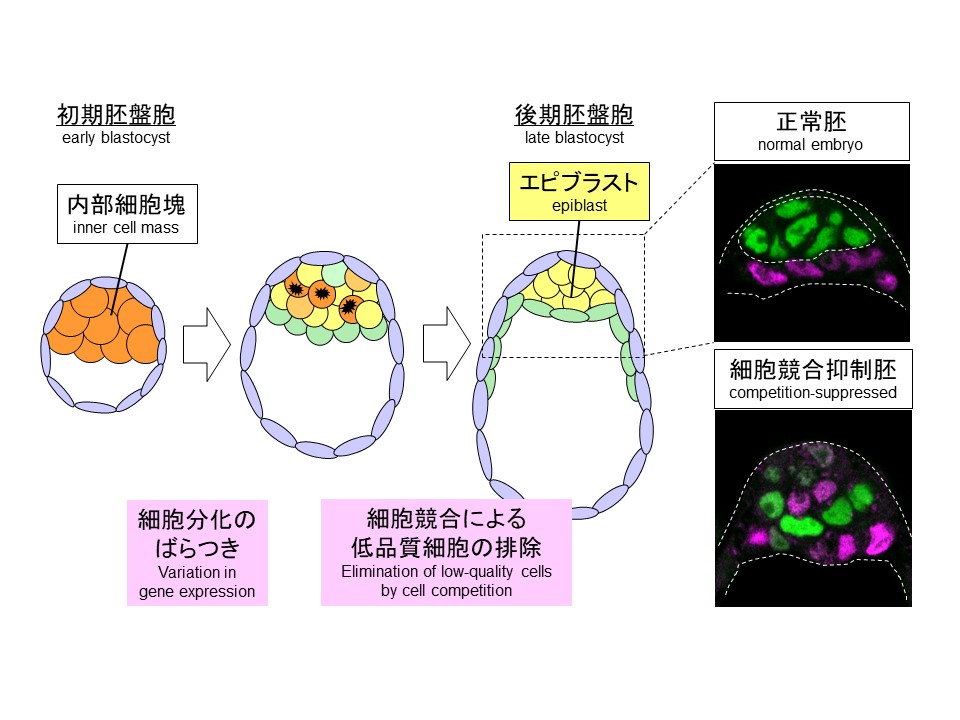
Cell competition in preimplantation mouse embryos. During formation of the pluripotent epiblast cells, cell competition functions as a quality control mechanism that eliminates low-quality cells formed by variation in gene expression (left). In competition-suppressed embryos, differentiation and allocation of cells are disturbed (right).
Members
| Hiroshi Sasaki (Professor) | sasaki.hiroshi.fbs[at]osaka-u.ac.jp |
|---|---|
| Masanori Uchikawa (Assistant Professor) | uchikawa.fbs[at]osaka-u.ac.jp |
| Hiromi Shimojo (Assistant Professor) | hshimojo.fbs[at]osaka-u.ac.jp |
| Toshihiro Aramaki(Assistant Professor) | aramaki.toshihiro.fbs[at]osaka-u.ac.jp |
You could probably reach more information of individual researchers by Research Map and researcher's search of Osaka-U.
- ※Change [at] to @
Q&A
- What is your hot research topic?
- The mechanisms and roles of cell competition in preimplantation embryos. We recently found that cell competition functions as a quality control mechanism of the forming of epiblast cells in preimplantation embryos, in which cell competition eliminates low-gene-expression/low-quality cells through apoptosis. We would like to reveal the mechanisms underlying this cell competition and also clarify the roles of this quality control in later embryogenesis and adult health.
- What is your breakthrough or research progress in the last 5 years?
- We identified the quality control mechanism though cell competition during the formation of the pluripotent epiblast in preimplantation mouse embryos. We have previously shown that Hippo signal-triggered cell competition takes place in cultured cells. It was not known whether similar cell competition also takes place during development, and if it does, what is its role? The epiblast formed in preimplantation embryos are pluripotent cells that will generate an entire embryo, and thus, all the epiblast cells should be of high quality. We recently found that strong variations are present among gene expression levels of the forming epiblast cells, and that cell competition acts as a quality control mechanism that eliminates low-quality (low pluripotency factor expressing) cells. This finding indicates that cell competition is a mechanism that supports correct development by overcoming variations in gene expressions during cell differentiation in development, and therefore, this is an identification of novel mechanism that supports the correctness of development.
- What kind of background do your lab members have?
- Most lab members have backgrounds in developmental biology. Some members have backgrounds in cell biology, microdevices and pharmacology.
- Do you collaborate with other institutions and universities?
- We collaborate with other laboratories when required to progress in our research or when other labs propose collaboration.
- What kind of careers do your Lab's alumni go on to?
- Academia and companies
- How do you develop your research?
- We found that one of the mechanisms supporting correct development is a quality control via cell competition in preimplantation embryos. As we need to reveal the mechanisms and the long-term effects of this quality control, it is important to clarify whether such quality control through cell competition is a general mechanism during cell differentiation in development or not. Furthermore, the mechanisms that support correctness of development should contain various intercellular communication mechanisms other than cell competition. Identification of such mechanisms are an important direction to be explored.
Research Highlights
Publications (Research Articles, Reviews, Books)
2020
The absence of SOX2 in the anterior foregut alters the esophagus into trachea and bronchi in both epithelial and mesenchymal components
Biol. Open 9, bio048728 2020 (PMID:31988094 DOI:10.1242/bio.048728)
Nodal paralogues underlie distinct mechanisms for visceral left-right asymmetry in reptiles and mammals.
Nat. Ecol. Evol. 4(2):261_269 2020 (PMID:31907383 DOI:10.1038/s41559-019-1072-2)
Sox2 gene regulation via the D1 enhancer in embryonic neural tube and neural crest by the combined action of SOX2 and ZIC2.
Genes Cells 25(4):242-256 2020 (PMID:31997540 DOI:10.1111/gtc.12753)
2019
Epiblast formation by TEAD-YAP-dependent expression of pluripotency factors and competitive elimination of unspecified cells
Dev. Cell 50(2):139-154.e5 2019 (PMID:31204175 DOI:10.1016/j.devcel.2019.05.024)
2018
Obesity in Yap transgenic mice is associated with TAZ downregulation.
Biochem. Biophys. Res. Commun. 505(3):951-957 2018 (PMID:30309656 DOI:10.1016/j.bbrc.2018.10.037)
Cooperation of Sall4 and Sox8 transcription factors in the regulation of the chicken Sox3 gene during otic placode development.
Dev. Growth Diff. 60(3):133-145 2018 (PMID:29520762 DOI:10.1111/dgd.12427)
Nasal and otic placode specific regulation of Sox2 involves both activation by Sox-Sall4 synergism and multiple repression mechanisms.
Dev. Biol. 433(1):61-74 2018 (PMID:29137924 DOI:10.1016/j.ydbio.2017.11.005)
2017
Neural Progenitor Cells Undergoing Yap/Tead-Mediated Enhanced Self-Renewal Form Heterotopias More Easily in the Diencephalon than in the Telencephalon.
Neurochem. Res. 43(1):180-189 2017 (PMID:28879493 DOI:10.1007/s11064-017-2390-x)
Notch and Hippo signaling converge on Strawberry Notch 1 (Sbno1) to synergistically activate Cdx2 during specification of the trophectoderm.
Sci Rep 7:46135 2017 (PMID:28401892 DOI:10.1038/srep46135)
Enhancer Analyses Using Chicken Embryo Electroporation.
Methods in Molecular Biology 1650:191-202 2017 (PMID:28809022 DOI:10.1007/978-1-4939-7216-6_12)
A Wnt5 Activity Asymmetry and Intercellular Signaling via PCP Proteins Polarize Node Cells for Left-Right Symmetry Breaking.
Dev. Cell 40(5):439-452 2017 (PMID:28292423 DOI:10.1016/j.devcel.2017.02.010)
2016
Roles and regulations of Hippo signaling during preimplantation mouse development.
Dev. Growth Diff. 59, 12-20 2016 (PMID:28035666 DOI:10.1111/dgd.12335)
Somatic cell reprogramming-free generation of genetically modified pigs.
Sci. Adv. 2(9)e1600803 2016 (PMID:27652340 DOI:10.1126/sciadv.1600803)
Electroporation of the Cas9 protein/sgRNA into early pronuclear zygotes generates non-mosaic mutants in mouse
Dev. Biol. 418(1):1-9 2016 (PMID:27474397 DOI:10.1016/j.ydbio.2016.07.017)
GFRA2 Identifies Cardiac Progenitors and Mediates Cardiomyocyte Differentiation in a RET-Independent Signaling Pathway
Cell Reports 16, 1-13 2016 (PMID:27396331 DOI:10.1016/j.celrep.2016.06.050)
Our ideal candidate (as a graduate student)
We are looking for a highly motivated person to work on our research topics as our lab member. Our lab welcomes the person who loves taking care of creatures, hand working and handcraft too. Any kind of background (such as your expertise or major) is available.
Contact
Laboratory for Embryogenesis, Graduate School of Frontier Biosciences, Osaka University,
1-3 Yamadaoka, Suita, Osaka 565-0871 Japan.
E-mail: sasaki.hiroshi.fbs[at]osaka-u.ac.jp (Prof. Hiroshi Sasaki)
- ※Change [at] to @
